- Home•
- Categories•
- Product Information•
- Attachments•
- Fiber Technology•
- TriLor® High Performance Polymer
Attachments
TriLor is a millable fiber-reinforced resin material designed for suprastructures for full arch restorations and bridge frameworks.
This super-strong resin flexes and bends under stress, transferring less energy to the implants, making it an ideal support material for implant restorations.
The TriLor Arch uniquely allows the dental professional to design the framework using a lathe or handpiece.

Customer Reviews:
Jeremy Wohlers, CDT
“Predictable outcome is predictable income. I use TriLor as a tool to make my implant retained cases predictable.”
Eugene Royzengurt
“TriLor has improved workflow in my laboratory and chairside while still achieving a very aesthetic outcome.”
TriLor® Arch Forms
TriLor® Arch allows the analog creation of connection structures between implants that can be embedded in removable prosthetic devices, as well as frameworks for transferring the position of the implants into the mouth, or simple reinforcement bars for a total definitive prosthesis.
TriLor® Pucks
A high-performance techno-polymer matrix with multi-directional glass fiber reinforcement for permanent, esthetic restorations on implants. TriLor exhibits natural flex and load parameters, making it the ideal replacement for unforgiving metal and zirconia frameworks.
TriLor® Indications & Material Comparison
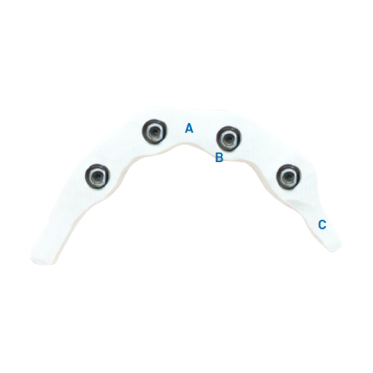
For Suprastructure Design
A. Minimum must be 7mm2 (3.5mm x 2mm)
B. Minimal Thickness around Cylinders is 0.8mm
C. Maximum Cantilever is 10mm

Zero toxicity, no sensitivity, no irritants
TriLor was tested as “zero sensitivity” according to all ISO 10993 and FDA testing standards:
- ISO 10993-3 — non-mutagenic Tests for genotoxicity, carcinogenicity and reproductive toxicity
- ISO 10993-5 — does not induce cytoxicity Tests for in vitro cytotoxicity
- ISO 10993-6 — non- irritant Tests for local effects after implantation
- ISO 10993 -10 — non-sensitizer test for irritation and skin sensitization
- ISO 10993-11 — no adverse phycal symptoms after injection (test of systemic toxicity)